Course Chapters
Section Tests
Online Calculators Linear Least Squares Regression Newton's Method Equation Solver
Related Information Links
|
Problem 3 Solution
Solution Steps for Part (a):If a sample of methane contains 1565 atoms, how much carbon and hydrogen are present?
Answer:This is different from the last problem because we are given the total number of atoms instead of the amount present of one of them. What do we know?
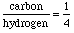 . .Let's cross multiply this proportion as usual to get:
So, we can substitute 4 carbons in for the hydrogen in the first equation:
Put the carbons together: (4 and 1 makes 5)
So what's left must be hydrogen: 1565 - 313 = 1252. Solution Steps for Part (b):Can a sample contain 1566 atoms?
Answer:Look at the work from part (a): We know that the total number of atoms must be divisible by 5.Think about what a ratio tells you when you can't have fractional parts of the items: The items must come in groups of 5 -- 1 carbon for every 4 hydrogens in this case. Using this idea, there can't be 1566 atoms because 1566 is not divisible by 5 because 5 carbon = 1565(total). Try it on your calculator; do you get a whole number? No! |
 Shodor
Shodorin cooperation with the Department of Chemistry,
The University of North Carolina at Chapel Hill
Copyright © 1996-2008 Shodor
Please direct questions and comments about this page to
WebMaster@shodor.org