| |
|
Dry Adiabatic Process
Knowing the rate at which rising air cools is vital in determining the
stability of the atmosphere. We have briefly introduced dry, moist, and
saturated adiabatic processes in previous readings, but because these
concepts are so important to the discussion on atmospheric stability, we
will take a few minutes to review them, as well as introduce a couple of
others that are of equal importance.
The Dry Adiabatic Process
When a parcel of air rises, it expands, and the temperature decreases.
Likewise, when air sinks, it compresses, and the temperature increases.
This phenomenon occurs without adding or withdrawing energy from the parcel
and is illustrated by the equation of state and Poisson's equation. When a parcel of
air, either dry or containing water vapor, rises or sinks without the addition or
extraction of heat, that process is said to be a dry adiabatic
process. Even though a parcel of air may contain moisture, if the parcel
is rising, then it cools according to the dry adiabatic lapse rate
until it reaches the dew point temperature
(Td).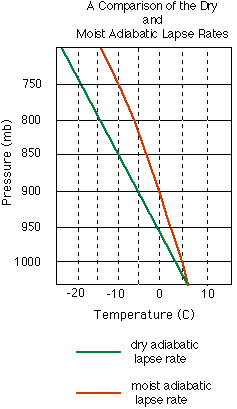 (Note: The dew point temperature for a
rising air parcel is not equal to the dew point temperature of the same
parcel at the surface. As the parcel rises, the dew point temperature
decreases slightly in response to the decrease in pressure.) We refer to
the pressure where the actual temperature equals the dew point temperature
as the Lifting Condensation Level
(LCL). At the LCL, the cooling process becomes a moist or saturated
adiabatic process. (Note: The dew point temperature for a
rising air parcel is not equal to the dew point temperature of the same
parcel at the surface. As the parcel rises, the dew point temperature
decreases slightly in response to the decrease in pressure.) We refer to
the pressure where the actual temperature equals the dew point temperature
as the Lifting Condensation Level
(LCL). At the LCL, the cooling process becomes a moist or saturated
adiabatic process.
Confused? Have a question? If so, check out the Frequently Asked Questions (FAQ) page or send mail to the OS411 tutor (os411tutor@shodor.org) with your question!
Report technical/content problems here
|
|